The Story Behind the Research: A Spotlight on Dr. Paola Leone
Rare Disease Day is about the people who turn hope into action. This month, we highlight the work of Dr. Paola Leone, a leading global authority on Canavan disease and a pivotal scientific collaborator for Myrtelle. While her roots are in genetic brain diseases, her name has become synonymous with the modern push for a Canavan disease cure.
A Legacy of Dedication
Dr. Leone has dedicated over 25 years to developing gene therapies for neurodegenerative disorders. Her “bench-to-bedside” approach was forged in the 1990s at Yale, where she collaborated on the first-ever human gene therapy for a genetic brain disease.
The Turning Point
Her impact on the Canavan disease community reached a new height in 2017. Recognizing that early trials needed to better target the white matter of the brain, she partnered with affected families to develop a “next-generation” therapy. This breakthrough focused on oligodendrocytes, which are the specific cells responsible for myelin. This strategic shift from general brain delivery to cell-specific targeting is now the foundation of Myrtelle’s lead gene therapy candidate, MYR-101.
“The strength of the Canavan community is what drives the science. We are here to bring the breakthroughs of the lab directly to the families who need them.” — Dr. Paola Leone
2025 in Review: A Landmark Year for MYR-101
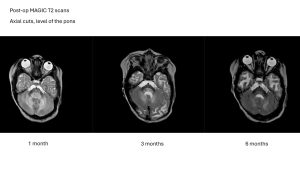
As we look back at the past year, the momentum toward a treatment has never been stronger. 2025 was a year of massive strides:
FDA START Pilot:
Myrtelle’s inclusion in the FDA’s START pilot program has streamlined our regulatory path, allowing for more frequent communication with the FDA to accelerate the commercial delivery of the MYR-101.
Nature Medicine Publication:
In September 2025, our landmark study proving that MYR-101 successfully restored myelin and reduced toxic NAA levels in children was published.
Meeting on the Mesa:
We presented at the Meeting on the Mesa encouraging interim results from our Phase 1/2 clinical trial where the MYR-101 has demonstrated a favorable safety profile with no serious adverse events related to treatment.
ASGCT Presentation:
Myrtelle presented insights on being one of four gene therapy companies selected for FDA’s START Pilot Program and the accelerated pathway for rAAV-Olig001-ASPA development.
Looking Ahead: ASGCT 2026
The momentum continues this year at the American Society of Gene & Cell Therapy (ASGCT) conference. Dr. Leone is expected to present:
Long-term Follow-up Data:
Updated results from the Phase 1/2 trial of MYR-101, focusing on safety and durable efficacy.
Regulatory Progress:
Insights into the path toward FDA approval, anticipated sometime this year.
Platform Expansion:
How this targeting technology can be applied to other leukodystrophies and monogenic disorders of the white matter.
How You Can Get Involved This Rare Disease Day
Awareness leads to action. Here is how you can support the Canavan community this month:
Share the Science:
Help us amplify the results of the Nature Medicine study to show the world the tangible progress being made.
Light Up for Rare:
On February 28, join the “Global Chain of Lights” by lighting up your home or office in Rare Disease Day colors (pink, green, blue, and purple).
Advocate for Research:
Support the foundations that provide the grassroots funding necessary for “early-stage” research that traditional grants often overlook.
Support Organizations that Support Patient Care:
Research is only one side of the coin. Organizations like the Cure Canavan Fund, the Canavan Foundation, and NTSD provide direct support for specialized physical therapy, essential medical equipment, and family counseling.
Together, we are making the “rare” seen, heard, and healed.

