
Where Newborn Screening Stands Today
Every baby born in the United States receives a newborn screening, which uses a few drops of blood from a heel prick to test for a panel of serious but treatable conditions. What it does not yet include, in any state, is Canavan disease.Families currently learn they may be at risk through carrier screening. It is a test that must be requested, most often by couples who already understand that they should ask, or by those who have already lost a child. For infants where testing is delayed, the diagnostic gap is irreversible. And while there is pre- and post-natal testing, it remains opt-in, invisible to most families until it is too late.
We Have Seen This Before. SMA Changed Everything.
Spinal muscular atrophy (SMA) offers the clearest roadmap for what is possible. For a condition to be added to the federal Recommended Uniform Screening Panel (RUSP), a treatment or care standard must first exist. If a child must wait months to become symptomatic before embarking on the diagnostic odyssey, that time equates to disease progression that cannot be reversed.
In July 2018, the federal government added SMA to the Recommended Uniform Screening Panel (RUSP). Since then, advocates successfully encouraged the adoption of newborn SMA screening in all 50 states. Following the implementation of newborn screening, approximately sixfold more children with SMA were identified and became eligible for treatment. Cure SMA’s presymptomatic treatment dramatically shifted the natural history of SMA, with most patients meeting appropriate developmental milestones, delivering outcomes that were simply not possible before universal screening existed (PubMed Central). That is what we, at Myrtelle, are working toward for Canavan disease.
The New Era Is Coming for Newborn Screening
The broader landscape of newborn screening is also evolving rapidly. Advances in whole genome sequencing are expanding what is possible. The GUARDIAN study in New York is already screening up to 100,000 newborns for hundreds of conditions, and a 2026 Florida initiative aims to cover 750 or more treatable genetic diseases. For Canavan disease specifically, researchers have developed a promising LC-MS/MS-based method for detecting elevated NAA levels. The key Canavan disease biomarker, directly from dried blood spots, is the same sample used in standard newborn screening today. Notably, NAA testing through urine sampling is already a commonly used diagnostic for Canavan disease and represents another avenue that could be leveraged for newborn screening.
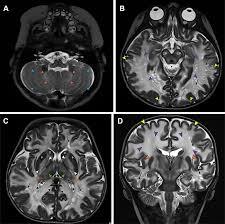
MRI imaging at 1, 3, and 6 months post-treatment shows progressive myelin growth following gene therapy.
Myrtelle’s Position and What Has to Happen Next
We are actively advancing each of these priorities. Through Myrtelle’s partnerships with the Cure Canavan Fund, the Canavan Foundation, and NTSAD, a robust community infrastructure is already in place. The science is progressing, the regulatory pathway is engaged, and commercial manufacturing is underway. The remaining step, establishing universal newborn screening for Canavan disease, represents the next critical frontier, and one we are committed to achieving.
Be Part of What Comes NextThe path from promising science to approved therapy to universal newborn screening doesn’t happen without a community behind it.
- Spread awareness. Most families don’t know to ask for Canavan carrier screening. Sharing this newsletter is a simple act with important outcomes.
- Support the organizations doing the daily work. The Cure Canavan Fund, the Canavan Foundation, and NTSAD are with families every step of the way.
- Follow our progress at myrtellegtx.com. We are in an important chapter right now, and an informed community is