Driven by a surge in novel scientific advancements, rare diseases have moved to the center of the national conversation. Researchers, policymakers, biotech leaders, and patient advocates are aligned on one uncomfortable truth: the system designed to get treatments to patients isn’t moving fast enough — and for families living with conditions like Canavan disease, that urgency is not theoretical.
At Myrtelle, we hear that urgency every day. And, we’ve spent years building the infrastructure to answer it.
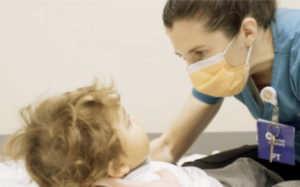
Navigating the FDA Moment
The FDA is under intense pressure right now. Staff reductions, shifting standards, and uncertainty are potential bottlenecks for rare disease treatments. The voices raising the alarm span both political parties and every corner of the biotech world.
For Canavan families, delays aren’t measured in months. They’re measured in milestones. That is precisely why Myrtelle’s position matters.
Myrtelle was one of seven companies selected for the FDA’s START Pilot Program (3 from CDER and 4 from CBER). Our MYR-101 (Canavan disease treatment candidate) was one of the three gene therapies chosen based on its potential clinical benefit for a rare disease with unmet medical needs and the company’s ability to accelerate market application submission.
The program provides direct, ongoing communication with the FDA, enabling frequent and faster feedback and a streamlined path for MYR-101’s development.
Our RMAT designation adds another layer of structural advantage, allowing MYR-101 to be considered for rolling review, priority review, and accelerated approval once the BLA has been submitted.
When the regulatory environment is turbulent, preparation and access to the right programs make all the difference.

The Science Is Finally Matching the Urgency
Leading scientists are describing this moment as a turning point, when gene therapy, biomarkers, and cell-targeting technologies have advanced to the point where researchers can begin addressing the root causes of diseases that were once considered untreatable.
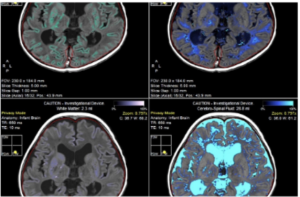
That is exactly what MYR-101 does. By precisely targeting oligodendrocytes, which are the cells responsible for myelin production, Myrtelle’s therapy addresses the source of Canavan disease rather than managing its symptoms.
The results published in Nature Medicine in September 2025 confirmed that MYR-101 restored myelin and reduced toxic NAA levels in treated children.
These findings represent an important step toward developing a disease-modifying therapy for Canavan disease. And critically, this platform isn’t limited to Canavan disease.
The same oligodendrocyte-targeting approach is being developed for Pelizaeus-Merzbacher Disease (PMD), H-ABC (Hypomyelination with Atrophy of the Basal Ganglia and Cerebellum), and Multiple System Atrophy (MSA). Multiple conditions, one powerful scientific platform.
The Economics of Getting There
Even the best science doesn’t reach patients without the right commercial infrastructure.
Industry leaders have been candid this year about the economics of rare disease: treatments cost hundreds of millions of dollars to develop, insurance systems are fragmented, and the path from approval to patient access is rarely straightforward.
Myrtelle has developed its commercialization strategy with this reality firmly in mind. Commercial-stage manufacturing is already underway, meaning that when regulatory approval comes, we are ready to deliver. And our relationships with the Cure Canavan Fund, the Canavan Foundation, and NTSD (National Tay-Sachs & Allied Diseases) ensure that the community infrastructure exists to support families through every step of that journey.
Stay With Us
Share the Science. The Nature Medicine publication is one of the most important milestones in Canavan disease research. Help us get it in front of families who need to know a treatment is being developed.
Support the Community. The Cure Canavan Fund, the Canavan Foundation, and NTSD are on the ground with families every day.
Stay Connected. Follow our progress at myrtellegtx.com and on social media. Together, we are making the “rare” seen, heard, and healed.